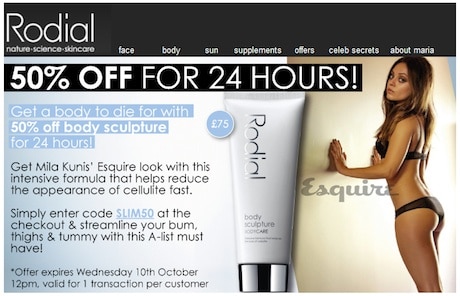
A beauty cream ad promising to give women the same body as Mila Kunis has been banned for being misleading.
Rodial failed to convince experts at the Advertising Standards Authority that its anti-cellulite product could really deliver the promised effect.
The email advertisement for a body sculpture cream was headlined ‘Get a body to die for with 50% off body sculpture for 24 hours’.
It featured an image of the 29-year-old actress, who starred in the 2010 film Black Swan, in her underwear and linked this to an Esquire magazine feature where she was described as ‘the sexiest woman alive’.
The ad went on: ‘Get Mila Kunis’ Esquire look with this intensive formula that helps reduce the appearance of cellulite fast’. Potential customers were urged to ‘streamline your bum, thighs & tummy with this A-list must have!’.
Rodial’s website says its products are the skincare choice for Angelina Jolie, Anne Hathaway, Gwyneth Paltrow and Rosie Huntington-Whiteley.
Defending its claims for the body sculpture cream, Rodial said it includes bio-marine actives, wheat protein and caffeine ‘to help break down stubborn fat and promote drainage and reduce fluid retention in the body’.
It also said the gel is ‘infused with pomegranate tannins to firm and tone your skin and boost collagen production’, which is associated with young skin.
The ASA said: ‘Most consumers would understand that the claim ‘‘streamline your bum, thighs & tummy with this A-list must have!’’ implied that consumers who used the product would be able to reduce the appearance of cellulite and tighten and smooth their bottom, thighs and tummy.
‘Evidence presented consisted of information about two active ingredients within the body sculpture product which made reference to in vivo trials on those ingredients. However, the trials themselves were not supplied. Because robust evidence was not presented to demonstrate the implied efficacy claims for the product or that Mila Kunis had achieved the look featured in the photo as a result of using the product, we concluded that the ad was misleading.’ The ASA said it should not be repeated.